To Put That Into Context
PSA-TRICOM is a vector-based therapeutic cancer vaccine regimen consisting of recombinant poxviruses containing transgenes from PSA and three T-cell costimulatory molecules. The vaccine was studied in a nonblinded phase II trial of patients with nonmetastatic CRPC and a rising PSA level, despite a castrate level of testosterone the participants were randomly assigned to either the vaccine or nilutamide. Patients were only required to have castrate levels of testosterone, to have undergone AAWD for 4 to 6 weeks, and to not be immunocompromised. Prior nilutamide therapy was an exclusion criterion. Nilutamide was given at a dosage of 300 mg daily for 1 month, followed by 150 mg daily subsequently. The vaccine was given as an initial priming dose of admixed recombinant vaccinia-based vaccine and then as a booster of recombinant fowlpox-based vaccine monthly. Subjects were followed every 3 months with CT and bone scans and with monthly PSA testing. Patients who developed rising PSA levels could cross over to the other arm. The time to treatment failure-defined as progression, secondary malignancy, or toxicity-was 9.9 months with the vaccine and 7.6 months with nilutamide . The median survival was 5.1 years for patients who received the vaccine vs 3.4 years for patients who received nilutamide alone . There were no grade 3 toxicities in the vaccine arm.
Orteronel
ODM-201
Special Situations And Drug Interactions
It is of vital importance to identify and differentiate the unalike clinical situations that we may encounter in our healthcare practice based on the baseline characteristics of the patients. Although we know that the chronological age does not always correspond to the real age of the patient, the tolerance in the elderly may differ from that of younger patients, with different metabolisms. In the same manner, it is important to take into account those patients with significant comorbidity or those who are polymedicated, in which certain nuances must be taken into account when prescribing these treatments .
Clinical Development Of Apalutamide: Phase I Ii And Iii Data
A total of 1207 patients were randomized in 2:1 fashion to receive apalutamide 240 mg daily or placebo and ADT was continued in both arms during the study. The patients were stratified by PSA-DT , use of bone-sparing agents and classification of nodal status as N0 or N1. The median age of the patients was 74 years , 71.3% had a PSA-DT 6 months, 10.0% used a bone-sparing agent, and 83.6% had N0 nodal status. Most of the patients had previously used a first-generation AR antagonist. At the planned primary analysis, apalutamide demonstrated significant improvement in MFS compared with placebo . Similarly, median PFS was 40.5 months in the apalutamide arm vs 14.7 months in the placebo arm . Of the other exploratory endpoints, apalutamide was associated with improved TTPP , time to symptomatic progression , and time to initiation of cytotoxic chemotherapy . PSA response rate was observed in 90% of patients in the apalutamide group as compared to 2% in the placebo group. The patients in the apalutamide arm reported stable overall health-related QoL as did the patients in the placebo arm. The second PFS was significantly longer in the apalutamide arm than the placebo arm . Of note, the OS data were not mature at the time of publication but apalutamide showed a trend toward improved OS with HR of 0.70. Notably, an exploratory landmark analysis in the SPARTAN trial suggested an association between MFS and OS in high-risk nmCRPC who develop metastases at 6, 9, and 12 months .39
Read Also: Can You Have Prostate Problems In Your 20s
Ethics Approval And Consent To Participate
The protocol and study materials were reviewed and approved by the Salus Institutional Review Board . All respondents in the qualitative interviews provided written informed consent prior to participating in the interviews. All respondents in the survey provided electronic agreement after reviewing the informed consent form electronically.
Other Resources Available To Patients With Pc

Participation in support groups can benefit patients with PC, enabling them to exchange experiences and techniques for improved medication adherence. Patients should be advised of additional resources provided by the treatment facility itself or through third parties . Onsite or online support groups and non-profit organizations can direct patients to sources of additional, factual information outside of their scheduled appointments with HCPs.
Read Also: Can You Remove A Prostate
Reviewimpact Of New Systemic Therapies In Overall Survival In Non
Three next generation antiandrogens significantly prolong overall survival in nm-CRPC
-
Mature survival data for these treatments are now available for meta-analysis
-
Simulated patient data-level may help to highlight differences among trials
-
Different study designs and follow-up may affect data interpretation
Index Patient : Symptomatic Mcrpc With Poor Performance Status And No Prior Docetaxel Chemotherapy
Clinical trials have generally excluded patients with a poor performance status from participation. Thus, most data regarding management of such patients is extrapolated from randomized trials of eligible patients who had a better performance status, as well as from some smaller trials and registries. Even a Phase 3 clinical trial that was presumptively designed for a population considered unfit for docetaxel still only allowed a performance status of ECOG 0-1. However, treatments with acceptable safety profiles do exist and should be considered, even in poor performance status patients. This is especially true in those patients in whom the poor performance status may be considered to be directly related to the cancer itself and thus whose status might improve with effective treatment. Treatments must be individually tailored in these patients after a careful discussion of risks and benefits with particular attention to patient QOL.
Guideline Statement 11
Clinicians may offer treatment with abiraterone plus prednisone or enzalutamide to patients with symptomatic, mCRPC with poor performance status and no prior docetaxel chemotherapy.
Discussion
Guideline Statement 12
Clinicians may offer treatment with ketoconazole plus steroid or radionuclide therapy to patients with symptomatic, mCRPC with poor performance status and no prior docetaxel chemotherapy who are unable or unwilling to receive abiraterone plus prednisone or enzalutamide.
Discussion
Don’t Miss: Does A Cystoscopy Look At The Prostate
Role Of Apalutamide Beyond Nmcrpc
Several trials are currently underway to evaluate the role of apalutamide in early and advanced stages of prostate cancer. During ASCO 2019 meeting, the results of the TITAN trial were presented. TITAN was a Phase III trial which assessed the efficacy of apalutamide plus ADT versus placebo plus ADT in patients with metastatic hormone-sensitive prostate cancer. The patients who previously received docetaxel for advanced prostate cancer were also enrolled. The primary endpoints were radiologic progression-free survival and OS. At the time of first interim analysis, apalutamide plus ADT was associated with improved rPFS as compared to placebo plus ADT .57 OS survival data were not mature at the time of the first interim analysis. Grade 34 adverse effects were 42.2% in the apalutamide plus ADT arm vs 40.8% in the placebo plus ADT arm.57
ATLAS, a Phase III, randomized double-blind placebo-controlled trial is evaluating the role of apalutamide in combination with GnRH agonist compared with GnRH agonist alone in patients with high risk, localized or locally advanced prostate cancer who are receiving radiation therapy as initial definite therapy.63 The primary endpoint is MFS.
|
Table 1 Phase III trials of apalutamide in prostate cancer |
Dont Miss: Prostate Cancer Types And Treatment
Integrating The Aua Guidelines Into Your Practice
Michael S. Cookson, MD, MMHC, describes how urologists can use American Urological Association guidelines to better manage metastatic castration resistant prostate cancer patients. He stresses the importance of urologists serving as the primary caregiver, the multidisciplinary care model, and organized sequencing of treatments and therapeutics.
You May Like: How To Prevent Prostate Problems Naturally
Recap: Recent Updates In The Treatment Of Nonmetastatic Castration
Aaron Berger, MD, discussed updates in the treatment of nonmetastatic castration-resistant prostate cancer.
In a recent OncView discussion, Aaron Berger, MD, vice president and chief medical officer at Associated Urological Specialists in Chicago, Illinois, shared clinical experiences and perspectives regarding treatments of patients with nonmetastatic castration-resistant prostate cancer .
Clinicians need to be aware of all the treatment options available in this space, as many FDA-approved indications have emerged in the past few years, Berger said.
Thereve been several new options for nonmetastatic castration-resistant prostate cancer to come to market, he noted. The first was enzalutamide followed shortly thereafter by apalutamide , and then most recently darolutamide . Weve used all of them in our advanced prostate cancer clinic, and its certainly an improvement over the previous options as first-generation antiandrogen therapies.
Berger detailed his strategies for therapy selection in this patient population, including insights in baseline patient characteristics, clinical trial data, and toxicity profiles of each novel agent that guide his decisions.
Role Of The Nurse Specialist In Managing Adverse Drug Events
Patients with PC receiving ADT and AR inhibitors can experience AEs that may impact their QoL but may not think to report them during clinic visits. Oncology nurses play a pivotal role in the management of patients with PC by educating them on disease state, risk and likelihood of progression, respective therapeutic options and TEAEs along their individualized treatment path. Nurse specialists may participate in treatment strategy, for example, collaborating with oncologists and urologists, on the possible benefits of a temporary dose reduction or interruption, or choosing the most appropriate treatment option for individual patients depending on their comorbidities and associated comedications. Shared decision-making strategies include patient education and referrals to resources for self-care. Effective and early management of TEAEs is likely to improve medication adherence and reduce discontinuation of therapy. Permanent discontinuation due to AEs can be an indication of treatment tolerability. In SPARTAN and PROSPER, the incidences of discontinuations due to AEs had increased between the primary and final analyses ., , , In contrast, in the final analysis of ARAMIS, the incidence of discontinuations due to AEs was unchanged from the primary analysis, remaining at 8.9% with darolutamide and 8.7% with placebo.,
Also Check: What Does The Prostate Gland Do For The Body
Cardiovascular Aes And Hypertension
Cardiovascular AEs have been linked to AR-targeted therapy and were identified as the most frequently reported AE that led to death in the PROSPER trial of enzalutamide. Therefore, patients should be monitored carefully for symptoms of cardiovascular disease at baseline and throughout treatment. Patients should also be educated on a heart-healthy diet and on how to recognize, control and prevent hypertension, deep vein thrombosis and pulmonary embolism. While the extent to which exercise can ease the potential metabolic consequences of ADT is uncertain, engaging in both aerobic and resistance exercises remains sound advice for all patients receiving ADT.
Luciana Saboya Brito Dal Col
1Universidade Estadual de Campinas, CampinasSP, Brasil, UroScience, Universidade Estadual de Campinas – Unicamp, Campinas, SP, Brasil
2Pontifícia Universidade Católica de Campinas, CampinasSP, Brasil, Pontifícia Universidade Católica de Campinas – PUC-Campinas, Campinas, SP, Brasil
Don’t Miss: Can Prostate Cancer Be A Secondary Cancer
Understanding Nonmetastatic Crpc: An Overview
Transcript:
Judd W. Moul, MD: Nonmetastatic castrate-resistant prostate cancer is prostate cancer that is no longer responding to hormone therapy, but the patient does not have metastases. Stepping back a little bit, lets first talk about castrate-resistant prostate cancer, or CRPC, which we used to refer to as hormone-refractory prostate cancer. Its basically a gentleman in our practice who has been on hormone therapy or androgen deprivation therapy and now has a rising PSA or clinical progression of the disease. He also has to have a low testosterone level, classically less than 50 ng/dL or even less than 20 ng/dL. His PSA has to be rising.
Nonmetastatic castrate-resistant prostate cancer is common, although its not crystal clear how common it is. I did some recent literature searches trying to answer that question. One European paper I found said that in the European Union, potentially 4% to 7% of prostate cancer patients who are living would have nonmetastatic castrate-resistant prostate cancer. Then I looked at data from the United States, and recently the American Cancer Society suggested there may be some 20 million, or up to 29 million, menwhich seems crazy highliving in the United States who are survivors of prostate cancer.
Transcript Edited for Clarity
The Role Of Genetic Testing For Inherited Prostate Cancer Risk
Dr. Veda N. Giri, MD, commenced the Philadelphia Prostate Cancer Consensus 2017 by explaining the purpose and need for creating updated, centralized guidelines regarding genetic testing for inherited prostate cancer. Dr. Giri urged the conference members to focus on addressing the criteria for referring a patient, how genetic counseling is relevant to men at risk for prostate cancer, which genes to test, and how this informs the management of patients.
Recommended Reading: Can Stress Affect Your Prostate
Oncology Nurses And Patients As Partners In Cancer Care
Oncology nurses can provide vital assistance to patients who may be incapable of processing and retaining all information conveyed to them during an initial encounter and who may prefer to maintain an active role in their treatment journey., Another important role of nurse specialists throughout the therapeutic course is to provide patients with reliable, factual information on what to expect from their prescribed treatment regimen, including TEAEs and life expectancy. Patients should be encouraged to ask questions if any aspects of their diagnosis, case management or prognosis remain unclear to them after initial meetings with their HCP.,
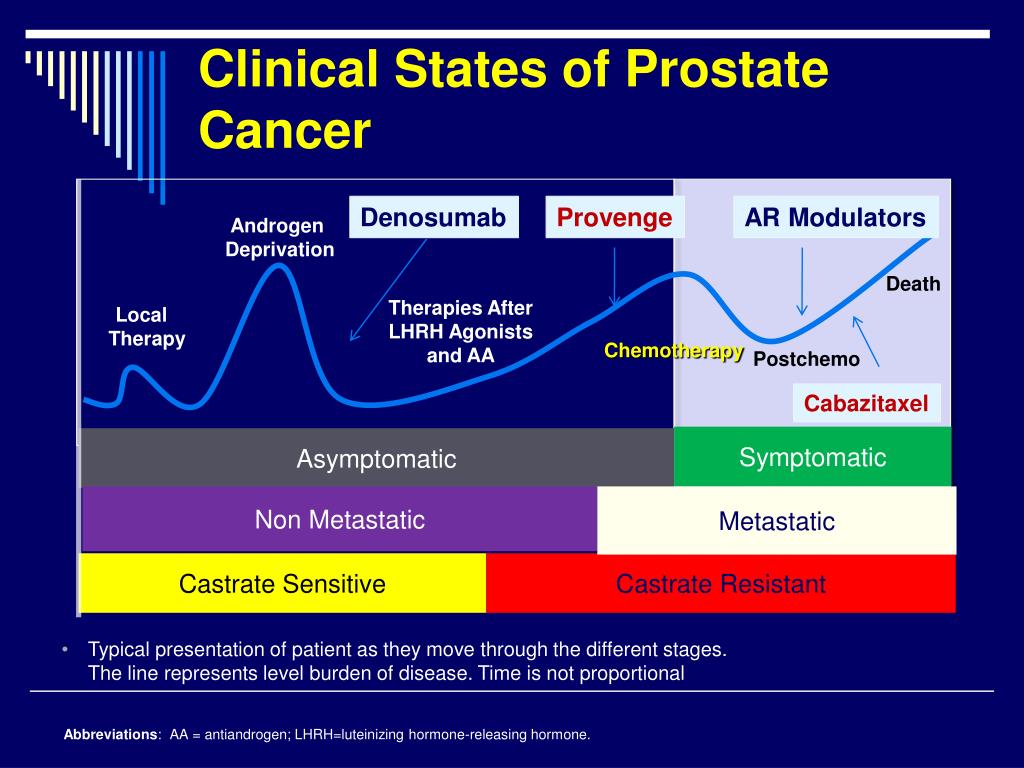
Index Patient : Asymptomatic Non
One of the first clinical presentations of CRPC occurs in a patient with a rising PSA despite medical or surgical castration. This is typically defined as a patient with a rising PSA and no radiologic evidence of metastatic prostate cancer. The Prostate Cancer Clinical Trials Working Group 2 defines PSA only failure as a rising PSA that is greater than 2ng/mL higher than the nadir the rise has to be at least 25% over nadir, and the rise has to be confirmed by a second PSA at least three weeks later. In addition, the patient is required to have castrate levels of testosterone and no radiographic evidence of metastatic disease.16 These patients represent a relatively common clinical presentation and the earliest clinical manifestation of castration resistance.
Guideline Statement 1
Clinicians offer apalutamide or enzalutamide with continued androgen deprivation to patients with non-metastatic CRPC at high risk for developing metastatic disease.
Discussion
Enzalutamide: Enzalutamide is a novel AR signaling inhibitor. It is a competitive inhibitor of androgen binding and also inhibits nuclear translocation of the AR, DNA binding and coactivator recruitment.18 This drug binds AR with a five- to eight-fold higher affinity than bicalutamide.18
Guideline Statement 2
Clinicians may recommend observation with continued androgen deprivation to patients with non-metastatic CRPC at high risk for developing metastatic disease who do not want or cannot have one of the standard therapies.
Also Check: Can Biking Cause Prostate Problems
M0 Space And The Spartan Trial
Eric J. Small, MD, defines nonmetastatic castration-resistant prostate cancer, or M0 disease. He then discusses the design, implementation, and results of the SPARTAN trial, which he also presented at the 2018 Genitourinary Cancers Symposium. The study tested the efficacy of apalutamide in men with M0 disease.
Future Treatment Landscape In Nmcrpc
Regarding unmet needs in the treatment space, Berger said that guidance for prescribers on drug-drug interactions is lacking. There are a lot of medications patients are on, whether its antihypertensives, diabetes medications, or cardiovascular medications, especially the anticoagulants that may have some interactions with these medications. And the guidance, as far as what we can glean from the studies, is not always clear about whats safe and what may not be safe, he said.
Another consideration is whether nmCRPC will continue to be a disease state in the future, as next-generation imaging techniques become more prevalent in the treatment landscape and reveal metastasis in patients who would have been formerly considered nonmetastatic.
When you have a scan that can pick up an area of metastasis at 0.2 to 0.3 ng/mL, it may turn out that these patients are metastatic. All of these studies were done with conventional imaging, Berger said. The big question as far as this entire disease state is, will it still be a disease state 5 years from now?
Overall, Berger said clinicians shouldnt shy away from prescribing these medications to their patients, given their tolerability and ease of administration. I would not be afraid of these medications because you can easily add them into your clinical practice without a lot of trepidation, he said.
Recommended Reading: How To Examine The Prostate
How Prostate Cancer Is Treated
In cancer care, different types of doctorsincluding medical oncologists, surgeons, and radiation oncologistsoften work together to create an overall treatment plan that may combine different types of treatments to treat the cancer. This is called a multidisciplinary team. Cancer care teams include a variety of other health care professionals, such as palliative care experts, physician assistants, nurse practitioners, oncology nurses, social workers, pharmacists, counselors, dietitians, physical therapists, and others.
The common types of treatments used for prostate cancer are described below. Your care plan may also include treatment for symptoms and side effects, an important part of cancer care.
Treatment options and recommendations depend on several factors, including the type and stage of cancer, possible side effects, and the patients preferences and overall health.
Cancer treatment can affect older adults in different ways. More information on the specific effects of surgery, chemotherapy, and radiation therapy on older patients can be found another section of this website.
Because most prostate cancers are found in the early stages when they are growing slowly, you usually do not have to rush to make treatment decisions. During this time, it is important to talk with your doctor about the risks and benefits of all your treatment options and when treatment should begin. This discussion should also address the current state of the cancer:
