Who Can Get Benefit From This Treatment
Most patients with prostate cancer undergo surgery or radiation therapy. Despite optimal therapy, many patients have relapses. Further, as a rule, they can be in for a long time to control with hormone therapy. Subsequently, in many patients, the tumor cells become insensitive to it and, therefore, should be administered chemotherapy. This often leads to side effects and reduces the patients status of life. In addition, tumor cells also are resistant to this treatment.
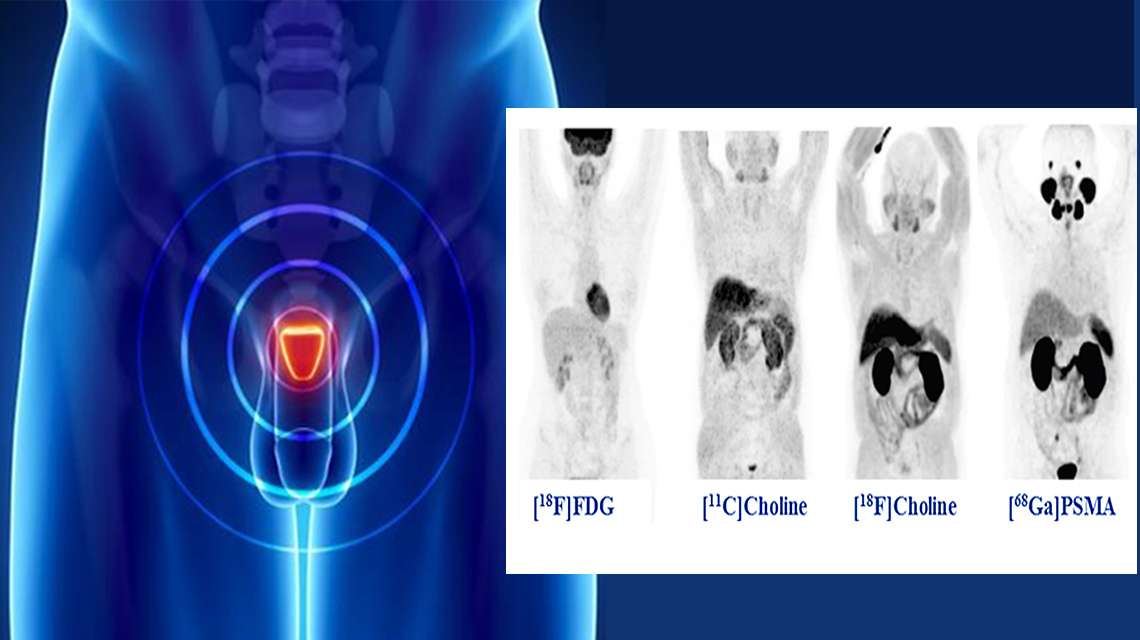
In this situation, we have the opportunity to administer 177Lu-PSMA as a new method of treatment. Treatment is performed only if metastases or a tumor can accumulate PSMA. To determine this, positron-emission tomography with PSMA-labeled 68Ga is performed.
Expertise In Lymphoma And Other Blood Cancers
Our doctors have helped discover and develop treatments now used to treat adult leukemia, lymphoma, multiple myeloma, myelodysplastic syndromes, and other blood cancers. They are nationally and internationally recognized leaders in the field. We know there are many subtypes of leukemia, lymphoma, and multiple myeloma your physician carefully assesses your disease using the most advanced diagnostic and molecular profiling techniques and considers this information, along with your overall health, to choose the most effective treatment. If you need a stem cell transplant, youll benefit from our experience performing hundreds of these procedures each year. We also have special expertise in transplants from mismatched donors and offer approaches that reduce the risk of complications.
- Bone marrow/stem cell transplantation. For some people with a blood cancer that is at high risk of coming back, has already come back, or can no longer be treated successfully with other therapies, stem cell transplantation may be an option. We perform hundreds of stem cell transplants a year. We have advanced transplant units offering individual monitoring and special airflow systems for patients with weakened immune systems. We also have special expertise in transplants from mismatched donors and offer approaches that reduce the risk of complications.
The Royal Marsden To Start Offering Lutetium Psma Therapy To Treat Advanced Prostate Cancer
Patients with advanced metastatic castration resistant prostate cancer to be offered an innovative molecular therapy which precisely targets cancerous cells reducing the exposure to the rest of the body.
Lutetium-177 PSMA is an innovative therapy used to treat metastatic castration resistant prostate cancer. The therapy works to reduce the tumour size and prevents the tumour from increasing, whilst also helping to improve the symptoms that these tumours might cause.
PSMA is naturally found on the surface of prostate cells in someone with prostate cancer there is an increase of PSMA expression. If the prostate cancer has spread to other parts of the body the PSMA will also be present in those areas. When administered intravenously the Lutetium-177 PSMA ligand will travel to those areas where the PSMA is present and emits radiation that will destroy the cancer cells the treatment is targeted to the cancer with very little radiation exposure to other parts of the body.
The Royal Marsden will offer PSMA therapy, which will be administered by an expert multi-disciplinary team. Nuclear Medicine consultants, Urologist consultants, specialised Nuclear Medicine nurses, Ward nurses, Nuclear Medicine Technologists, and Nuclear Medicine Physics team will work together to ensure the best treatment and care.
All the potential side effects and the necessary precautions will be carefully explained to patients during the clinic appointment with the Nuclear Medicine consultant.
Don’t Miss: Can You Get Prostate Cancer From Asbestos
A Leader In Liver Cancer Care
Our healthcare professionals in the Center for Liver Disease and Transplantation have exceptional experience diagnosing and treating benign and malignant liver tumors and liver metastases . Our surgeons perform minimally invasive liver cancer surgery whenever possible. If you have early-stage liver cancer, you may be able to have a liver transplant if not, you may have partial removal of liver tissue to remove your cancer. We also offer direct application of chemotherapy , intense heat , or radiation directly into a tumor interventional endoscopy to open blocked bile ducts targeted therapies radiation therapy and immunotherapy.
Biochemical And Clinical Responses
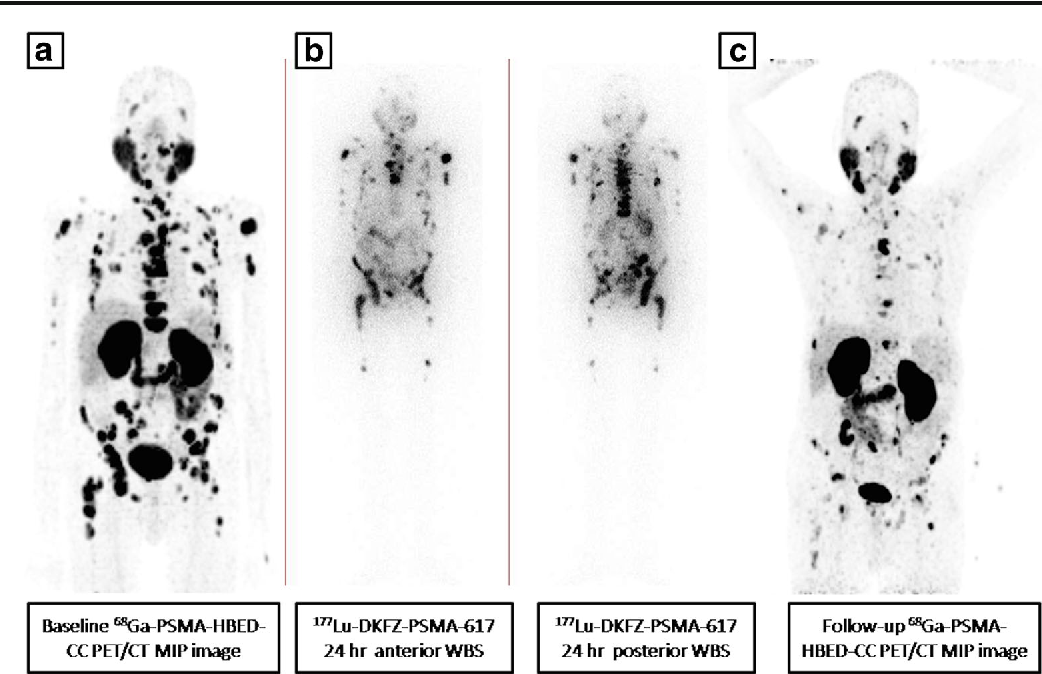
At a median of 8 weeks after the first cycle of 177Lu-PSMA-I& T RLT, PSA was declined in 26 patients and PR was achieved in 18 patients , indicating an improved efficacy for most patients. Furthermore, at the 8-week follow-up after the last cycle of therapy for all patients , the best PSA response showed that 28 patients had a PSA decline . Overall, 10 patients had PD , 25 patients had PR . The number of patients achieving maximum PSA decline of > 50% and > 90% were 22, 4, respectively. At follow-up, 10 patients had developed clinical progression and 5 were dead .
Figure 3 Waterfall plot of best PSA decline compared with baseline. Plots ordered from worst to best responder. Red dotted line represents 30% PSA decline. *The maximum PSA decline > 90%.
`Of the 40 patients, 35 patients had pain at baseline and the VAS decreased compared with baseline after all cycles . KPS, QOL were increased in 36 patients and were significantly improved after treatment .
Read Also: When To Get Your First Prostate Exam
Radiolabeling Stability And In
Radiolabeling of PSMA-617 with 161Tb wasachieved at specific activities up to 100 MBq/nmol at a radiochemical purity of98% .161Tb-PSMA-617 was stableover at least 1 h , but showed radiolytic degradation when incubated forlonger time periods. In the presence of l-ascorbic acid, 161Tb-PSMA-617 was stableup to 24 h and did not show any signs of radiolytic degradation. The determination of then-octanol/PBS distribution coefficient of 161Tb-PSMA-617 resulted in a value of3.9±0.1 .
Biodistribution Studies And Dose Estimation
Time-dependent biodistribution of161Tb-PSMA-617 was assessed in PC-3 PIP/flutumor-bearing mice and compared to the data previously obtained with177Lu-PSMA-617 . The uptake of161Tb-PSMA-617 in PC-3 PIP tumor xenografts reached amaximum at 4 h p.i. and decreased slowly over time . Accumulation of 161Tb-PSMA-617 inPC-3 flu tumors and other non-targeted organs was in the range of blood activitylevels or below at any evaluated time point. The radioligand was cleared via thekidneys over the first few hours after injection . These results confirmed that the tissue distributionprofile of 161Tb-PSMA-617 was equal to the data previously published for177Lu-PSMA-617 2) .
You May Like: Can Low Grade Prostate Cancer Become Aggressive
How Psma Lights Up Cancer Cells
In 2021, the U.S. Food and Drug Administration issued national approval to two new prostate cancer imaging tests based on similar technology. On a PET scan, the test lights up the cancerous cells that would otherwise be hidden, enabling doctors to precisely target treatment.
Both advances in imaging and therapy rely on targeting PSMA, which is not found on most normal cells but is overexpressed in cancer cells, especially those that have spread. The PSMA molecule was cloned at MSK in the early 1990s.
The Molecular Imaging and Therapy Service, led by Heiko Schöder, played a key role in the development and testing of a slightly different PSMA-directed imaging technology at MSK.
This advance is the result of years of work by the community of physicians promoting the use of PSMA agents, Dr. Schöder says. Its gratifying to see a collaborative effort result in a breakthrough that has the potential to make a difference for so many patients with advanced prostate cancer.
Before receiving the therapy, patients in the VISION trial were scanned with PSMA-directed PET imaging to make sure enough PSMA was present in the cells to make them likely to respond to the treatment. If so, they received the radioactive drug by injection over four to six sessions, spaced six weeks apart.
As a next step, Dr. Morris and colleagues are looking into using the PSMA-directed therapy earlier rather than only after the prostate cancer has spread.
Michael Morris
The Practice Of 225ac
Much of the clinical practice of 225Ac-PSMA TAT is derived from lessons learned from the clinical application of 177Lu-PSMA for PSMA-based radioligand therapy of mCRPC. Practice guidelines on the application of 177Lu-PSMA-617 for PRLT of mCRPC have been published by many professional societies and groups .
Don’t Miss: Signs And Symptoms Of Prostate Cancer In Males
What Causes Prostate Cancer
Prostate cancer is the most common form of cancer in men over the age of 50. Currently, researchers are unable to determine exactly what causes prostate cancer. DNA changes that are inherited or caused by certain lifestyle choices are believed to play a role in why some individuals develop cancer and others do not. It is believed that inherited genes account for about 5 to 10 percent of prostate cancers. Exposure to radiation, chronic inflammation, exposure to cancer-causing chemicals, high levels of androgens, or insulin-like growth factor-1 are also all believed to cause gene changes which lead to cancer.
Assessment Of Therapy Study
The efficacy of the radionuclide therapy was assessed by the tumorgrowth delay , which was calculated as the timerequired for the tumor volume to increase x-fold over the initial volume at Day 0.The tumor growth delay indexwas calculated as the TGDx ratio of treated mice overcontrol mice for a 2-fold and 5-fold increase of the initial tumor volume.Statistical analysis was performed by a one-way ANOVA with Tukeys multiplecomparison post-test using GraphPad Prism software . A value ofp< 0.05 was considered statisticallysignificant. The median survival was calculated by KaplanMeier curves usingGraphPad Prism software .
Potential early side-effects related to the exposure to radiationwere evaluated by the assessment of absolute and relative organ weights, selected clinical chemistry plasma parameters including creatinine, blood urea nitrogen , alkaline phosphatase , total bilirubin, and albumin , as well as via histological analysis of bone marrow andsalivary glands. The data were analyzed for statistical significance .
Read Also: How To Fix Enlarged Prostate
Is This Treatment Safe
The radiation used in the Lutetium-177 is designed to only destroy the cancer cells. With theranostic, treatment becomes more personalized so that cancer and its metastasis are effectively pinpointed but the whole body is never exposed to the radiation. Only the cancer cells are irradiated and destroyed. Blood tests will be performed to makes sure that the radiation is not damaging healthy tissues. Imaging tests are also performed to ensure that the radioactive material at the tumor sites has been correctly absorbed.
Side effects of the therapy may include:
- Dry mouth
- A brief decline in production of blood cells
Hyperthermia Therapy Supercharges Treatment
Hyperthermia is the application of heat to parts of the body to increase blood flow and enhance the efficient delivery of oxygen and nutrients. It also super-charges & enhances our low-dose chemotherapy protocol.
For decades weve witnessed the miraculous benefits of I-Therm as part of a comprehensive cancer treatment program cancerous tumors shrink pain is alleviated neurological conditions improve and healing occurs more rapidly.
Don’t Miss: How To Massage Your Own Prostate
Look For These Symptoms
We encourage all our male patients, particularly those over the age of 50, to be aware of potential dysfunction of their prostate gland. Be aware of:
- trouble urinating
- general pain in the lower back, hips or thighs
- pain when urinating
- pain when ejaculating
If you should experience any of these, you should schedule an appointment with one of our practitioners immediately.
Are You Searching For Possible Treatment Options For Metastatic Prostate Cancer
Prostate Cancer is the most common form of cancer diagnosed in men. While most patients respond well to surgery or radiation treatment, some develop advanced disease and become incurable. Prostate cancer is the second leading cause of cancer deaths in men.
The American Cancer Society estimates that over 30,000 new cases of prostate cancer were diagnosed in 2020 alone. Prostate cancer is usually treated by various methods including Surgery , radiation therapy, hormonal therapy, chemotherapy, immunotherapy as well as targeted therapy which has been recently added to the list. In the early stages of cancer, these methods might give long-term remission but in advanced metastatic prostate cancer, they only help in shrinking the tumors instead of curing the disease.
There are two main types of treatment that are commonly used in advanced metastatic castration-resistant prostate cancer i.e. chemotherapy and radiation therapy. Both methods not only kill the cancer cells but also affect the nearby healthy tissues and therefore, are not very effective against prostate cancer because they don’t target the cancer cells only.
Read Also: Why Does My Prostate Hurt
Current Evidence For The Efficacy Of 225ac
The first set of studies demonstrating the efficacy of 225Ac-PSMA in the treatment of mCRPC came from Heidelberg, Germany. A report of two patients who had exhausted available conventional therapies, one of whom was deemed ineligible for 177Lu-PSMA due to diffuse red marrow metastases of mCRPC and the other experienced disease progression on 177Lu-PSMA therapy, was the first to show the efficacy of 225Ac-PSMA-617 in large volume metastases of mCRPC . In the patient with diffuse marrow metastases of mCRPC, normalization of serum PSA and 68 Ga-PSMA PET/CT imaging findings occurred after four cycles of 225Ac-PSMA-617 with no significant change in hematologic indices. In the other patient with radioresistant to beta-emitting radionuclide therapy with 177Lu-PSMA therapy, a large volume of peritoneal metastases invading into the liver resolved, and serum PSA dropped to below detectable limit after three cycles of 225Ac-PSMA-617 . This report provided the first preliminary insights into the capability of 225Ac-PSMA TAT for mCRPC, including its safety and efficacy in the setting of diffuse red marrow metastases, its effectiveness in the setting of radioresistant to PRLT with a beta-emitting radionuclide, its ability to eradicate large volume metastases, and its efficacy as a last-line therapy in patients who have failed multiple lines of therapy for mCRPC.
Fig. 2
How You Get It
PSMA therapy usually takes place in a hospitalâs nuclear medicine department. A health care professional injects the medicine into a vein in your arm.
It takes about 30 minutes for the drug to infuse into your bloodstream.
You get anti-nausea meds and a diuretic to help flush the Lu 177 from your system. After that, you may need to wait in the hospital for a few hours or longer to make sure your radiation levels start to come down.
A day or two after your treatment, you’ll have an imaging test called a single-photon emission computerized tomography scan. This checks to make sure your therapy hit the right targets.
You May Like: What Is Super Beta Prostate
Impacts Of Baseline Parameters On Therapeutic Efficacy
Patients were classified into a remission and non-remission, and progression and non-progression groups according to PR and PD. Univariate logistic regression analysis of pretherapeutic signs and prior treatments confirmed that higher BMI was related to clinical progression . We also found that higher baseline serum AST, alkaline phosphatase , and lactate dehydrogenase were associated with poorer outcomes 6.91 . In addition, baseline imaging parameters including post-therapy SPECT/CT , pre-therapeutic 68Ga-PSMA-11 PET-CT showed no significant correlation with response .
Table 4 Univariate logistic regression analysis on basic factors associated with therapeutic effect.
Table 5 Univariate logistic regression analysis on basic imaging factors associated with therapeutic effect.
We assessed the relation among AST, ALP, and LDH with multivariate logistic regression analysis . Only AST and BMI were therefore included in the multivariate logistic regression analysis, and both were independent predictors of progression after RLT .
Figure 7 Scatter diagram showing correlation between three baseline serum biomarkers. AST and ALP, AST and LDH, and ALP and LDH.
Table 6 Multivariate logistic regression analysis on factors associated with disease progression after PRLT.
Patient Selection For 225ac
The typical patient selected to undergo TAT with 225Ac-PSMA will be a patient with histologically confirmed prostate cancer whose disease has become castration-resistant and has progressed on conventional therapies. The decision to treat a patient with 225Ac-PSMA should be made in a multidisciplinary setting where the disease history, prior therapies, patients comorbid conditions and health state, available alternative treatments, and the patients wishes are thoroughly discussed. After this discussion, the multidisciplinary team must agree that 225Ac-PSMA therapy is the most suitable therapy option for the patient. Based on the known toxicity of 225Ac-PSMA therapy, sufficient vital organ reserve, especially of the bone marrow and kidneys, must be present before submitting a patient to TAT with 225Ac-PSMA. Acceptable organ reserve commonly applied in routine clinical practice is as follows:
-
Bone marrow function: hemoglobin level> 8 g/dL platelet count> 75×109/L, white cell count> 3×109/L
-
Renal function: serum creatinine< 2 times the upper limit of normal
Recommended Reading: Prognosis For Prostate Cancer That Has Spread To Lymph Nodes
Side Effects And Toxicity Of Prlt
There were no transfusion-related complications or treatment-related deaths within 24 h after 177Lu-PSMA-I& T treatment, and no significant changes was seen in heart rate, blood pressure, or body temperature. Chest tightness with panic and shortness of breath occurred in one patient within 1-2 months after PSMA RLT treatment. Grade 1-2 fatigue and drowsiness were seen in 3 and 2 patients, respectively, on the second day after therapy. About 6 patients had mild to moderate xerostomia during follow-up.
Grade 1-4 impairment of bone marrow dysfunction occurred in up to 28 patients . Before RLT, 5 and 2 patients had grade 2 and 3 anaemia, respectively, 8 had grade 1 leukopenia, and 1 had grade 1 and grade 3 thrombocytopenia. After treatment, 5 patients reported grade 3-4 anaemia, 1 patient reported grade 3 leukopenia, and 5 patients had grade 34 thrombocytopenia. In all 86 cycles, there was a significant decrease in haemoglobin , erythrocyte counts , leukocyte counts and platelet counts .
No significant hepatotoxicity or nephrotoxicity was reported in the study. Aspartate aminotransferase and alanine aminotransferase levels remained relatively stable after each cycle, while 2 patients diagnosed with liver injury with elevated AST, 1 patient developed transient grade 1 renal impairment with a slight increase in serum creatinine after therapy. All adverse events are listed in Table 3.
Table 3 Treatment related toxicity occurring up to 12 weeks after treatment cessation.
What Are The Benefits Of Psma Pet Treatment At Ucsf
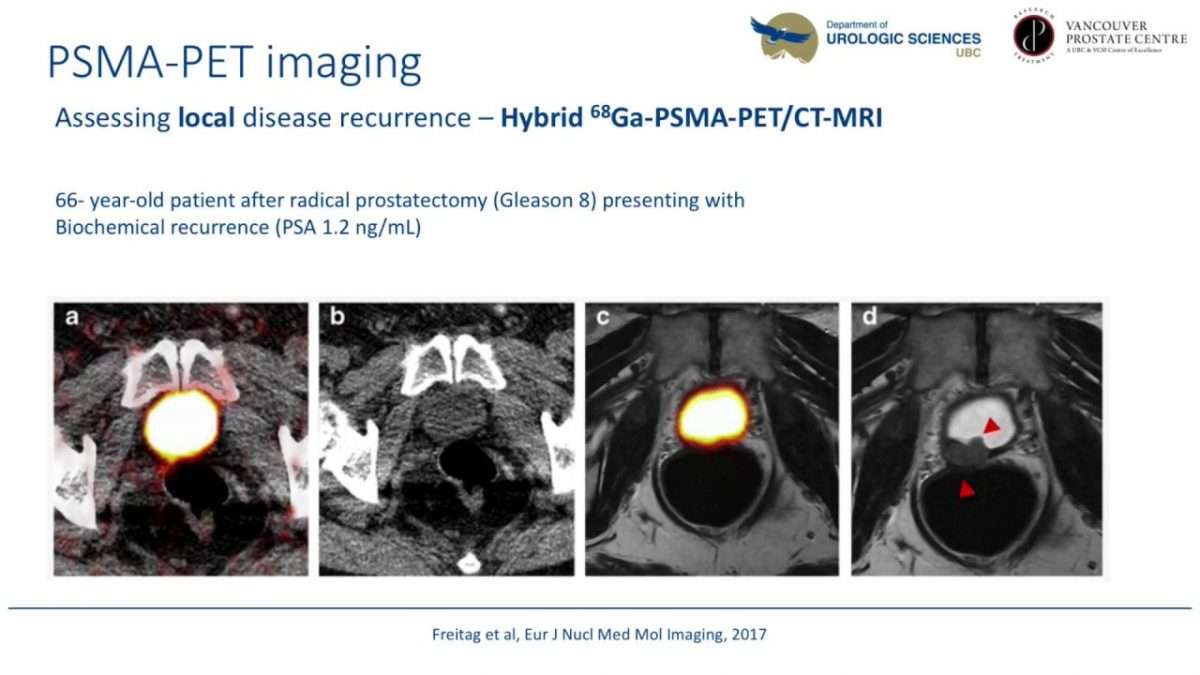
- FDA approved imaging technique for prostate cancer.
- The PSMA PET scan can identifiy cancer that is often missed by current standard-of-care imaging techniques.
- The PSMA tracer can also be used in conjunction with CT or MRI scans.
- UCSF is only one of two medical centers in the U.S. that offers the FDA approved PSMA PET.
- PSMA PET is more effective and precise for localizing mestatic prostate cancer.
- UCSF researchers, along with colleagues at UCLA, studied PSMA PET for several years to better precisely locate prostate cancer.
- PSMA PET works using a radioactive tracer, called 68Ga-PSMA-11, which is manufactured on site at UCSF.
Don’t Miss: What To Expect With Prostate Radiation
