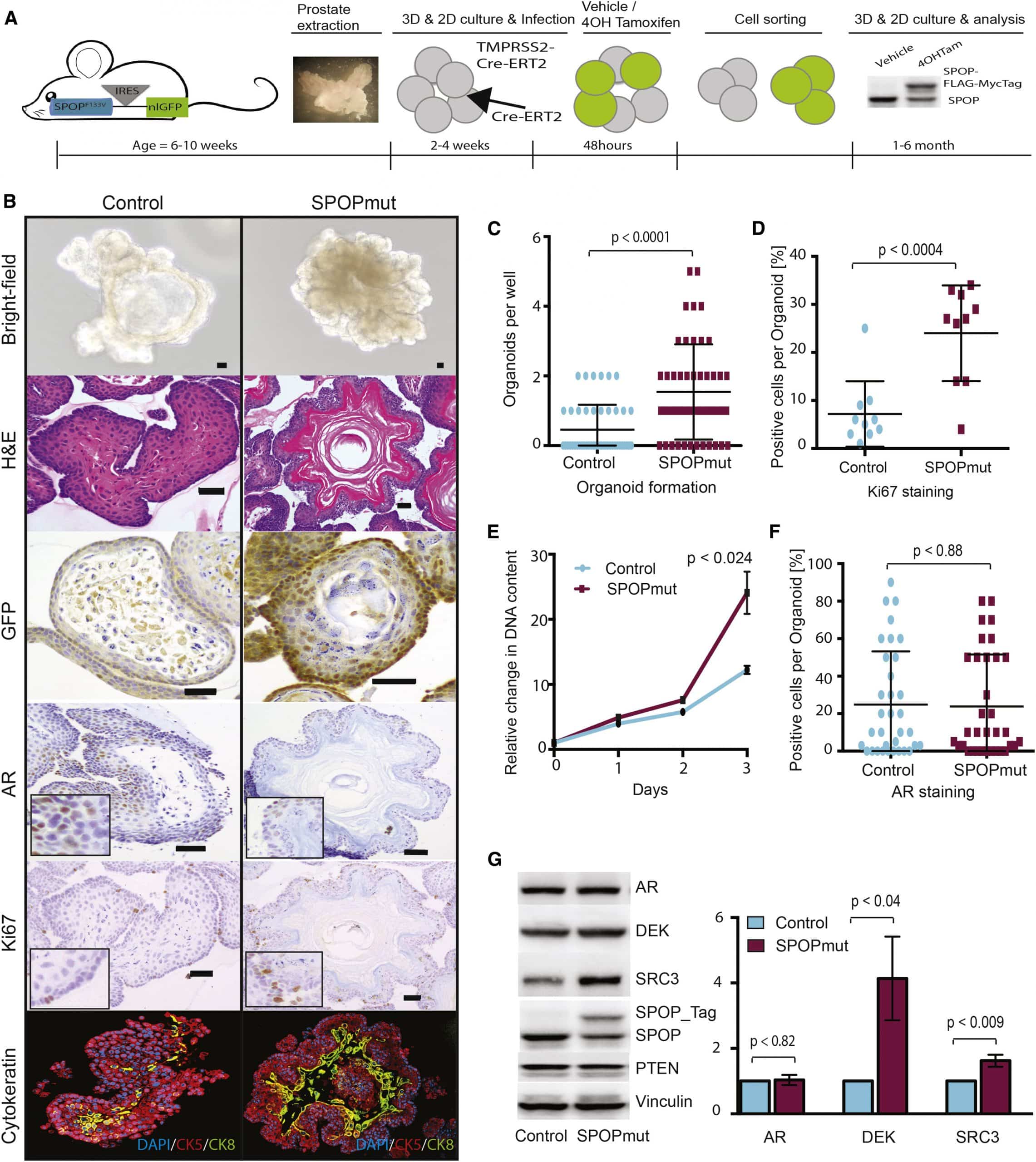
Nhprei And Bhprei Tissue Recombination Xenograft Model
To avoid issues with viral transformation, a series of spontaneously immortalized benign human prostate epithelial cell lines called NHPrE1 and BHPrE1 were generated . Although these cell lines lack AR expression in vitro, tissue recombination with inductive urogenital sinus mesenchyme induced luminal epithelial differentiation with AR expression. NHPrE1 cells were characterized as a progenitor line due to their high expression of CD133/CD44/OCT4/PTEN and the ability of as few as 10 cells to regenerate fully secretory glandular structures when recombined with inductive UGM. The BHPrEI cells were characterized as an intermediate cell line due to enhanced expression of p63/p53/p21/RB and the necessity to xenograft a minimum of 200,000 cells with inductive UGM to regenerate fully differentiated glandular structures. Genomic analysis detailed the small number of rearrangements and amplifications likely responsible for the immortalization. These cell lines represent a major advancement in our ability to accurately model transformation-independent effects of specific genes on cancer initiation. Future studies will assess the role of hormones and stroma, as well as oncogenes and tumor suppressor genes in carcinogenesis.
Rna Extraction For Rna Seq Analysis
RNA from FFPE mouse tumour tissue was extracted. Briefly, 2-4 sections were transferred into RNAase free microcentrifuge tube. The paraffin was removed by incubating in xylene and ethanol. For lysate and total RNA purification, digestion buffer and proteinase-K was added to the samples as per manufacturers instructions . The samples were spun briefly followed by transferring the supernatant to a new RNAase free microcentrifuge tube. The RNA containing tubes were incubated for 15 minutes at 80°C. The lysates were then passed through RNA purification microcolumn and centrifuged for 1 minute at 14,000 RPM. The microcolumns were washed according to manufacturers instructions and the RNA eluted using the Elution solution .
Prostate Carcinoma In Transgenic Lewis Rats
Laura E. Johnson, Jordan T. Becker, Jason A. Dubovsky, Brian M. Olson, Douglas G. McNeel
University of Wisconsin Carbone Cancer Center, Madison, WI 53705, USA
Corresponding to:
Abstract
Transgenic rodent models of prostate cancer have served as valuable preclinical models to evaluate novel treatments and understand malignant disease progression. In particular, a transgenic rat autochthonous model of prostate cancer using the SV40 large T antigen expressed under a prostate-specific probasin promoter was previously developed as a model of androgen-dependent prostate cancer . In the current report, we backcrossed this strain to the Lewis strain, an inbred rat strain better characterized for immunological analyses. We demonstrate that Lewis transgenic rats developed prostate adenocarcinomas with 100% penetrance by 25 weeks of age. Tumors were predominantly androgen-dependent, as castration prevented tumor growth in the majority of animals. Finally, we demonstrate that Lew-TRAP rats could be immunized with a DNA vaccine encoding a human prostate tumor antigen with the development of Lewis strain-specific T-cell responses. We propose that this Lew-TRAP strain, and prostate tumor cell lines derived from this strain, can be used as a future prostate cancer immunotherapy model.
Key words: Lewis rat transgenic prostate cancer vaccine
Submitted Nov 09, 2012. Accepted for publication Nov 16, 2012.
doi: 10.3978/j.issn.2304-3865.2012.11.06
You May Like: What Are The 5 Early Warning Signs Of Prostate Cancer
In Vitro Growth And Morphological Comparison Of E006aa
After establishing the E006AA-hT cell line in culture from a fast-growing tumor in NOG-SCID mouse, we performed its systematic characterization and comparison with its parental cell line, E006AA. PCR-based mycoplasma detection assays detected no mycoplasma contamination in the E006AA-hT cell line. As shown in Figure 1, they have a large fibroblastic morphology with long processes and at confluence form stream of cells . The E006AA-hT cell line grows slightly faster than the E006AA-Par cells . The population doubling time was found to be approximately 18 hr, slightly less than the parental E006AA . Like its isogenic parental cells, the E006AA-hT subline not only proliferated in serum-free medium, but also thrived in growth media with low serum concentration or under an androgen-depleted culture condition in charcoal-stripped FBS and at low cell density . The E006AA-hT cells can routinely grow in RPMI-1640 or DMEM supplemented with 0.5% or higher FBS. E006AA-hT cell line had very good recovery and viability when cultured from the cryopreserved state and was stable in a long-term culture beyond passage 40.
Figure 1
Erbb3 And Erbb4 And Autophagy
The role of ErbB3 in modulating autophagy is not well studied. In the prostate cancer cell line, LNCaP, it was found that neuregulin induces ErbB2/ErbB3 dimerization and activation, accompanied by autophagy and cell death exhibiting autophagic characteristics . Cell death induced by neuregulin is caspase-independent, but can be inhibited by 3-MA indicating the involvement of autophagy. The mechanism of neuregulin-induced autophagy involves ROS, JNK, and Beclin 1, but is independent of class I PI3K/Akt/mTOR1 pathway inhibition. Apparently, autophagy following neuregulin treatment is incomplete, which might explain the observed cell death .
Several ErbB3 inhibitors and pan-ErbB inhibitors, which inhibit ErbB3 and other family members, have been developed and are being tested as new tools for cancer therapy . Testing the effect of such inhibitors on autophagy modulation is worthwhile, given the role of ErbB3 in activating the PI3K/Akt/mTOR1 pathway . Currently, there is no evidence for cross-talk between ErbB4 and autophagy. However, a link between ErbB4 and autophagy is plausible, since ErbB4 also regulates the PI3K/Akt/mTOR1 pathway .
Recommended Reading: Can You Get Prostate Cancer From Masturbating
Analysis Of Immune Responses
Antigen-specific T-cell proliferation
Splenocytes harvested from immunized animals were evaluated using methods previously described . Specifically, 2Ã105 cells were cultured with 2 µg/mL human PAP protein , 2 µg/mL individual peptides , or 10 µg/mL phytohemaglutinin for 72 hours at 37 °C in an atmosphere of 5% CO2. Then, cultures were pulsed with 1 µM BrdU for eight to twelve hours. Antigen-specific, proliferating CD4+ or CD8+ T cells were detected using an intracellular flow cytometric staining method according to the manufacturerâs standard protocol, and BrdU incorporation was measured and analyzed as previously described .
Interferon gamma enzyme-linked immunosorbent assay
Splenocytes from immunized rats were cultured with the human PAP protein, individual peptides , or PHA for 72 hours as described above. The presence of IFNγ in the culture media was determined using a quantitative capture ELISA specific for rat IFNγ as described previously . Results are reported as the mean IFNγ concentration and standard deviation from multiple replicates.
Antigen-specific antibodies
The presence of antibodies specific to human PAP in the sera of immunized rats was determined by indirect ELISA, as described previously .
Tumor Surveillance By Gray
US imaging was performed using a portable sonography device . Prewarmed US coupling gel was applied to the abdominal wall before imaging. 2D images of the mouse prostate and adjacent anatomy were acquired using a linear-array transducer in B-mode with a center frequency of 11-MHz. The probe was positioned perpendicular to the abdominal wall in a sagittal orientation, and the bladder was identified as a round anechoic structure. The probe was moved until an echogenic tumor was identified on the dorsal side superior to the base of the bladder. Images of the bladder and tumor were acquired. Maximum sagittal length and height of the tumor was measured in the longitudinal plane. The probe was rotated to view the tumor in the transverse plane, and the width of the tumor was measured. Tumor volume was calculated by 0.52×maximum length×height×width.
Don’t Miss: How To Get A Free Prostate Cancer Badge
Pathology Of Specific Genetically Engineered Mouse Models
PTEN/AKT pathway.
With the increased realization that prostate cancer involves multiple signaling pathways, many studies have focused on engineering additional genetic lesions into the Pten null prostate cancer model to study their potential cooperation with Pten loss in prostate carcinogenesis. A number of such models were examined by the panel members. In several of the models, introduction of another genetic lesion led to more aggressive tumors.
Yu Chen has studied a model that overexpressed ERG in Pten null prostate cancer and found that in comparison with Pten loss alone, ERG significantly accelerated tumor development. Examination of tumors from this model revealed adenocarcinoma at 12 to 15 weeks of age and anaplastic invasive adenocarcinomas with sarcomatoid carcinomas by 25 to 30 weeks of age . Studies by Carver and colleagues had previously shown that ERG expression in a germline Pten+/â mice resulted in development of adenocarcinoma, whereas mice with heterozygous loss of Pten alone only developed mPIN.
More recently, Chen and colleagues have generated a mouse with homozygous deletion of the Lrf transcription factor along with homozygous deletion of the Pten gene. Histologic examination of a tumor from a 7-month-old mouse revealed sarcomatoid carcinoma similar to the mouse with Pten and p53 homozygous deletion described earlier, consistent with enhanced progression due to Lrf deletion .
MYC pathway.
ERG pathway.
Retinoblastoma pathway.
RAS/RAF/MAPK pathway.
Growth And Metastasis Profile Of B6cap Cells
Table 1: Metastasis profile of B6CaP allografts
|
Delivery |
| Other sites |
Lung , Lymph Node , Eye , Epididymis , Brain , Spleen |
B6CaP tumors were implanted by subcutaneous, intravenous, intracardiac, or orthotopic routes. Some subcutaneous tumors were resected when they reached 1 cm diameter. For comparison, MycCaP mice were injected into syngeneic mice by intravenous route. Columns represent number of mice with local growth at injection site, lung metastases, or bone metastases at necropsy. For mice with intracardiac B6CaP injections, lower panel indicates additional sites of metastases. Any Met refers to any distant metastasis, or growth at any site other than heart/pericardium.
Figure 3: Intracardiac injection of B6CaP cells generates widespread bone and soft tissue metastases. Representative images of metastases in femur, vertebrae with spinal cord compression, skull/jaw lung, adrenal gland, abdominal lymph node, seminal vesicle, Eye, brain, and spleen. Immunohistochemistry for pan-cytokeratin and AR. Bone metastases generated osteoblastic and osteolytic lesions . Bone lesions were occasionally large and exophytic . After intracardiac injection, median survival was 40 days with 21% of animals surviving to the 9-week endpoint . Venn diagrams representing the numbers of mice with metastases to bone, lung, or other non-lung soft tissue sites, total N = 44 . Venn diagram representing the locations of bone metastases to skull, spine, or limbs, total N = 34 .
You May Like: Is Cayenne Pepper Good For Your Prostate
Cell Line Generation And Maintenance
Mouse prostate epithelial cells were generated from dorsal, ventral and lateral lobes of the prostate from Probasin Cre-/- mice. Murine prostate cancer cells were generated from tumours derived from the dorsal, ventral and lateral prostate lobes of a trp53-/-/Pten-/- Pb-Cre4 mouse 7. Tissue was manually dissociated under sterile conditions and cell lines were generated as described in Cell Line Derivation and Supplementary Figure-1A.
The TRAMP-C1 murine prostate carcinoma cells were purchased from ATCC and maintained in DMEM high glucose medium, supplemented with 4mM L-Glutamine, 5% FBS, 5% Nu Serum, 0.005mg/ml of Bovine insulin, and 10nM Dehydroisoandrosterone . The MPEC and DVL3 cell lines were maintained in RPMI-media supplemented with 10% FBS, L-Glutamine and 100nM DHT.
Serum Psa Histological And Immunohistochemistry
Blood samples were collected from the retroorbital sinus before sacrifice. PSA levels were analyzed using an Electro-ChemiLuminescence ImmunoAssay . Orthotopic tumors were removed, fixed in 10% neutral-buffered formalin, and embedded in paraffin. Serial Sects. were cut on a microtome and mounted on glass slides. Sections were deparaffinized in Histoclear and hydrated in a graded series of alcohols and running tap water. Histopathology was performed using standard hematoxylin and eosin staining. Immunohistochemical staining was performed with a Vectastain ABC Elite kit . Briefly, sections were deparaffinized and hydrated, and endogenous peroxidase activity was blocked with 0.3% hydrogen peroxide in methanol for 20 min. Antigen retrieval was performed using the Antigen Unmasking Solution . Sections were cooled and rinsed in PBS, incubated in blocking solution for at least 30 min at room temperature, and incubated with primary antibodies for CD31, VEGF-A, Ki-67 , androgen receptor , PSA, p63 , or P504S overnight at 4 °C. Sections were incubated with the appropriate biotinylated secondary anti-goat, anti-mouse, or anti-rat immunoglobulin for 30 min at room temperature. The antigenantibody reaction was visualized using 3,3-diaminobenzidine tetrahydrochloride as a substrate. Sections were examined by light microscopy.
Also Check: How To Decalcify The Prostate
Dna Profile Analysis Of The E006aa
To prove the derivation of the E006AA-hT cell line, PCR-based powerplex DNA profiling was performed to amplify 16 STR loci , as shown in Table 1. This genomic DNA profile analysis, which examines highly polymorphic STR DNA loci, uniquely discriminates and identifies unrelated human cell lines. Amelogenin is used for gender identification. Both cell lines were compared against the most commonly used PCa cell lines including LNCaP, C4-2, C4-2B, PC-3, DU145, VCaP, 22RV1, 22Rv1, and MDA-PCa2b. There were 100% matches for allele 1 and 2 at the indicated DNA-loci between the E006AA-Par and E006AA-hT cell lines. RT-PCR analysis with species-specific -actin and GAPDH primers verified the human-derivation of E006AA-hT cell line .
Assays For Growth In Serum
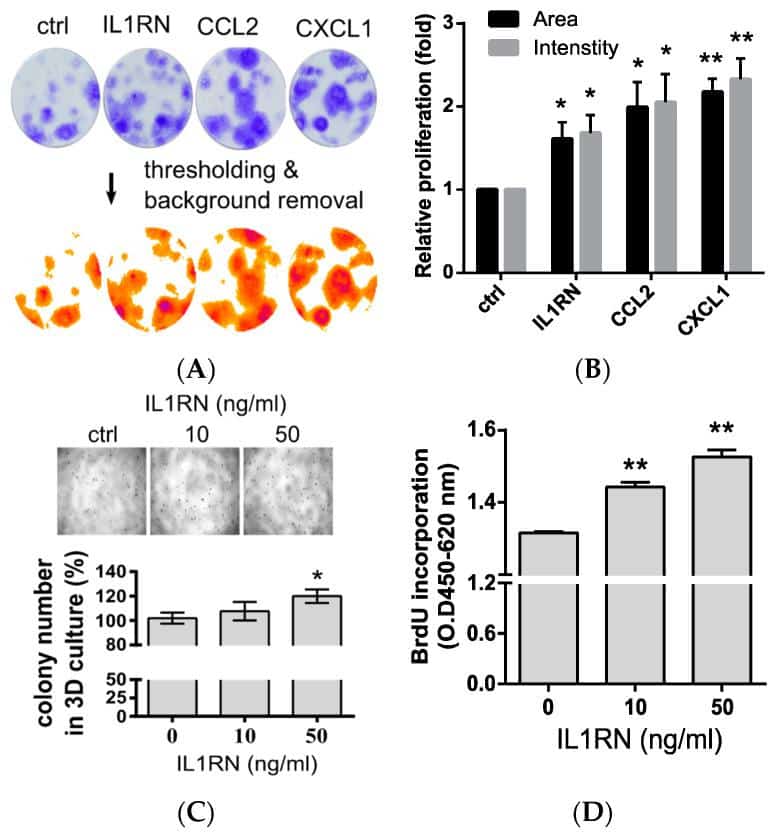
For testing the efficiency of growth of these cell lines in an androgen-free environment, a modified serum-free medium was used . It was composed of DMEM/F-12 supplemented with 0.5% glucose , and 2mM glutamine, 10g/ml insulin, 5.5 g/ml transferrin, 6.5 ng/ml selenium, and 6 ng/ml rhEGF . Initially, cells were plated in 6-well plates in the maintenance medium for overnight, washed two times with 1×PBS, and then cultured in SFM for 5 days. The culture medium was changed every 2 days, and the cell proliferation rate was determined at those time points by cell counting. SFM containing 1 or 5 nM of methyltrienolone , a synthetic androgen agonist, was utilized to determine the effect of androgen on cell proliferation.
Read Also: What Happens In A Prostate Exam
Enzalutamide Drug Screen And Gene Expression Associations With Response
We next performed drug screens on the cell line panel using enzalutamide. Consistent with previous reports, and similar to other hormone targeting agents in vitro, we found that the response as measured by GI50 were in the µM range . The response largely tracked with AR status , where responsive cells had high levels of AR protein expression and AR null cells were non-responsive. Similarly, cells with splice variants in AR were more resistant to enzalutamide. Surprisingly, there were several cell lines that were null for AR expression by both expression and Western analysis that were responsive to enzalutamide. This included the cell lines DU145, CA-HPV-10, and HH870.
Figure 3
Enzalutamide response in the prostate cancer cell line panel. Example dose response curves for a responsive and non-responsive prostate cancer cell line. Error bars are +/ standard deviation of triplicate measurements. GI50 values for each of the cell lines divides the samples into responsive and non-responsive clusters. Lower bars represent lower doses of drug required to inhibit growth by 50%. For non-responsive lines, GI50 values were set to maximal dose tested. GSEA plots for Estrogen and Androgen response elements show differences between enzalutamide responders and non-responders. The association is significant for ER associations but fails to reach significance for AR .
Allograft Establishment And Passage
Allograft tumors were minced and dissociated in DMEM/F12 media containing 10% Fetal Bovine Serum , Collagenase/Hyaluronidase mix at 37°C for 1 hour. 2.5 U/mL Dispase and 0.05 mg/ml DNAse I were added and cells were vigorously triturated for 1 min and passed through a 40 µm cell strainer with the aid of a syringe plunger. Cells were pelleted, washed twice, and resuspended in PBS. Live cells were counted with Trypan Blue dye and 1 × 10^6 viable cells in 100 µL PBS were injected subcutaneously in the shaved flank of C57BL/6J mice. Subcutaneous tumor size was measured with calipers. For intracardiac or intravenous inoculation, cells were similarly dissociated, and 1 × 10^5 viable cells in 100 µL PBS were injected via tail vein or by percutaneous intracardiac injection. For orthotopic inoculation, 1 × 10^5 cells in10 µL PBS were surgically injected into the anterior prostate lobe. To generate castration resistant cell lines, mice were surgically castrated when subcutaneous tumors measured 1 cm in greatest dimension. After complete regression, tumors were monitored weekly until regrowth began, which was considered castrate resistant recurrence.
Also Check: What Age Should You Get Your Prostate Checked
In Vitro Migration And Invasion Assays
The effect of AR silencing on cell migration was performed using 8-m transwell filters with modification as described previously . For the invasion assay, the upper compartment was coated with 50 g Matrigel to form a matrix barrier. A suspension of cells in medium containing 1% FBS and 0.1% BSA was added to the upper compartment. The lower compartment was filled with 400 l basal medium containing 10% FBS as chemoattractant. After 24 h for migration or 48 h for invasion, the non-migratory cells on the upper surface were removed by a cotton swab and the cells on the lower surface were fixed and stained with the Diff-Quick solution . Migrated or invaded cells in each transwell filter were counted from ten randomly chosen fields. The experiment was performed in quadruplicate and repeated three times independently.
> > > One Crazy Prostate Trick All Men Over 40 Should Try
Symptomatic treatment of an enlarged prostate usually involves a combination of medication and lifestyle changes. A diet rich in fruits and vegetables may be the best option if you suffer from chronic urination. It will help the body adjust to the increased size of the prostate. Also, taking regular urination intervals will help retrain the bladder to function properly. Inactivity also contributes to urine retention, and cold temperatures can increase the urge to urinate.
Invasive treatment of enlarged prostate includes medication that relieves the pressure on the urethra and bladder. However, if the condition is severe, it may require surgical intervention. If treatment is not successful, the enlarged prostate can become a potentially life-threatening disease. As the hormone levels in the body change, the enlarged prostate can lead to various complications, including urinary retention and even cancer. This is why it is critical to see a doctor for further evaluation.
You May Like: Prostate Hormone Treatment Side Effects
